|
This makes sense because there are about 14.01 grams of nitrogen for every 1 mol of nitrogen, using our ratios above. So all we have to do now is use the correct units, which would be grams per mole, or g/mol. It means when you have one mole of nitrogen, it is equivalent to about #14.01# grams of nitrogen, and it is also equivalent to about #6.02214 * 10^23# nitrogen atoms:ġ4.01 g N = 1 mol N atoms = #6.022 * 10^23# N atoms Nitrogen has an average atomic weight of 14.0067, so a mole of nitrogen gas is 28.0134 grams. 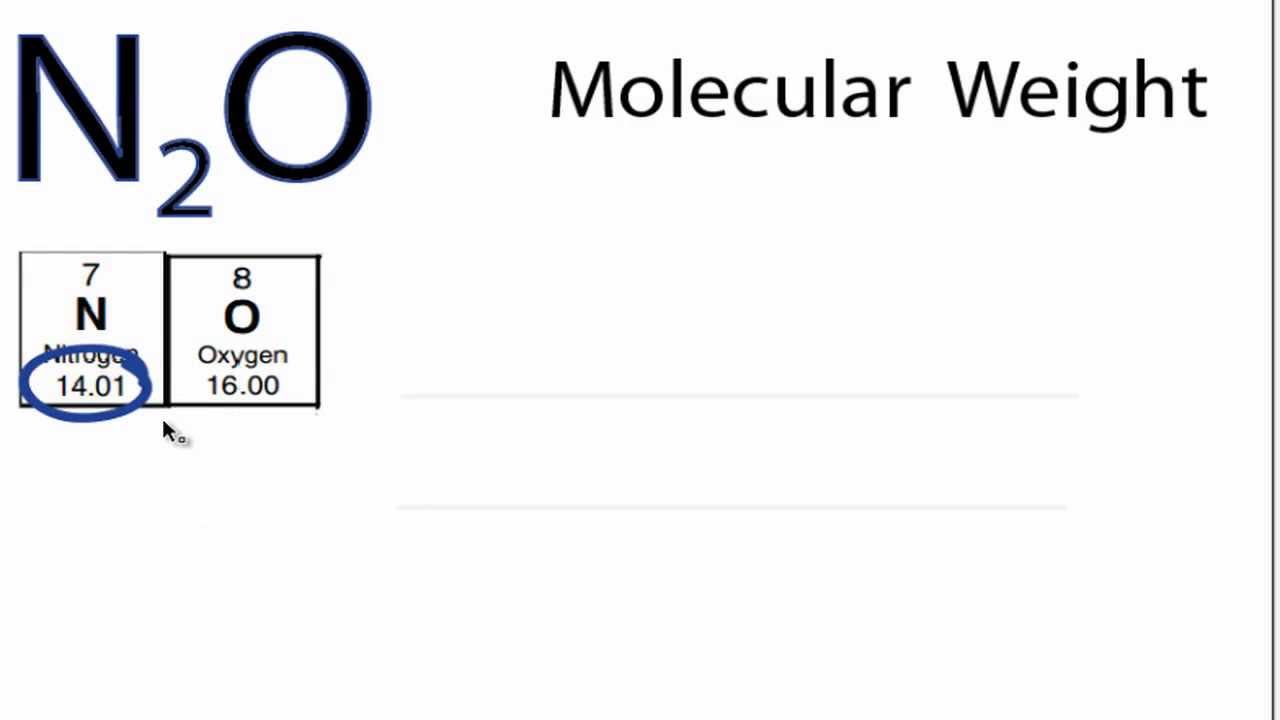
As nitrogen gas is composed of two nitrogen atoms, the formula mass of. Nitrogen gas has consists of pairs of nitrogen atoms (N2). See also our theoretical yield calculator for chemical reactions (probably your next stop to finish the problem set). Nitrogen has a atomic mass of 14.007 amu. The molar mass of NO (Nitrogen monoxide) is: 30.006 grams/mol. Numerically, this is the molar mass of nitrogen. Molar mass of nitrogen gas is 28.014 g/mol. Oxygen has a molar mass of 15.9994 g/mol and nitrogen has a molar mass of 14.0067 g/mol. Using a periodic table, we can find that nitrogen has an atomic mass of about #14.01# amu. This number, #6.02214 * 10^23#, is called Avogadro's number and is very useful when finding molar mass and converting between grams, moles, and particles. Numerically, it's the same as the element's atomic mass in units of amu (atomic mass units).Ī mole, or mol for short, is a quantity of any given thing that is equal to #6.02214 * 10^23# particles. Molar mass is the quantity of an element in grams for every one mole of atoms of that element. Molar mass of sodium 23.0 g / mol Similarly, one mol of ammonia contains 6.022 × 1 0 23 NH 3 molecules we Thus we can write Molar mass of NH 3 17.0 g / mol Number of moles (n) Molar mass of the substance Mass of substance in gram nple 16: Calculate mass of one atom of nitrogen in gram.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
